The Levy laboratory’s research efforts have led to key contributions towards unraveling cellular and molecular mechanisms for the resolution of lung inflammation, injury and infection. Our past work has focused on mediators of inflammation as the key molecular conveyors underlying the pathobiology of asthma and lung inflammation. Our work has also characterized that inflammatory conditions of the lung are also characterized by a deficiency of molecular mediators for the resolution of inflammation. Research projects in the laboratory are focused in the following areas:
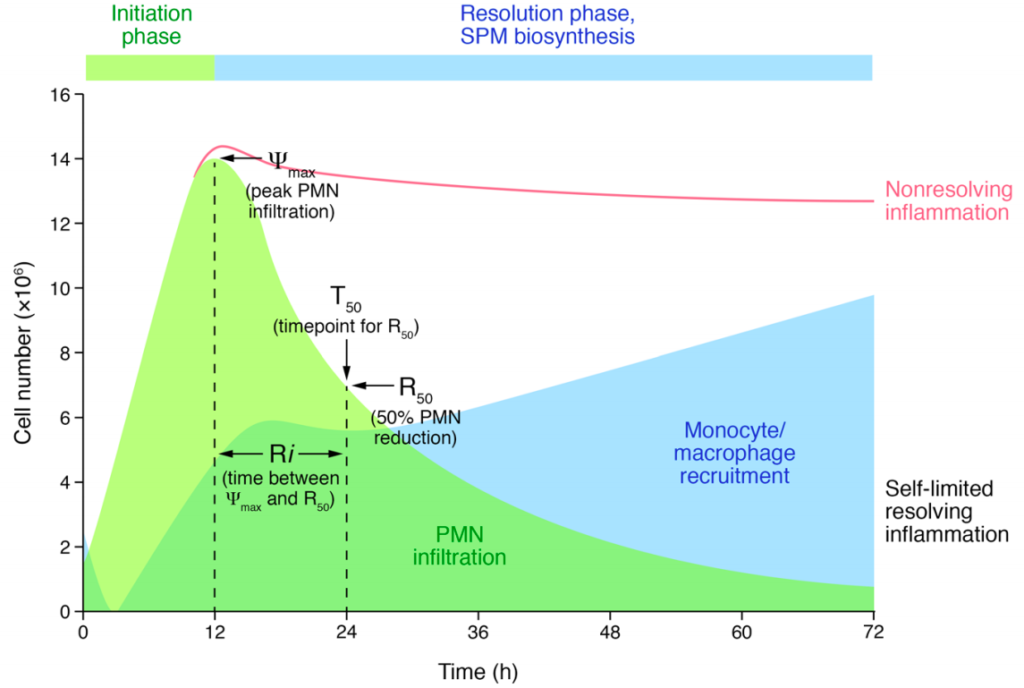
Resolution is an active process of inflammation.
With our collaborators, our investigations into the molecular and cellular mediators and mechanisms for the resolution of inflammation led to an appreciation that resolution is an active process. This was initially established for arachidonic acid metabolism in which the fate of this essential fatty acid switched from prostaglandins and leukotrienes (pro-inflammatory mediators formed early during the upstroke of inflammation) to lipoxins (counter-regulatory mediators (i.e., stop signals) formed late during resolution).
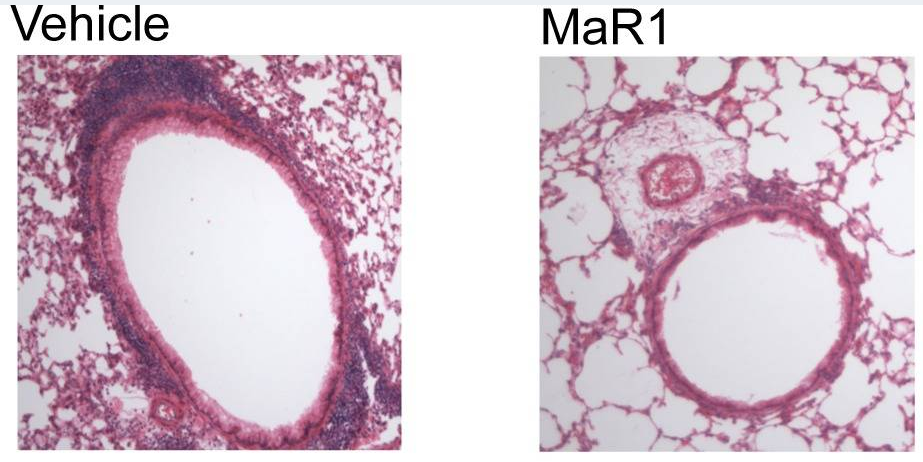
Specialized Pro-Resolving Mediators restrain lung inflammation and promote host defense
In addition to arachidonic acid conversion to lipoxins, the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are enzymatically transformed to stereoselective and cell-type specific mediators that promote resolution of lung inflammation from injury, infection and noxious stimuli (i.e., allergen). Our laboratory was the first to identify and characterize these mediators, namely resolvins, protectins and maresins – now collectively referred to as specialized pro-resolving mediators (SPMs) – in lung tissue responses. Current projects in this area include the study of the effects of SPMs on lung inflammation in murine models of disease (i.e. bacterial pneumonia, asthma, acid injury, viral infection).
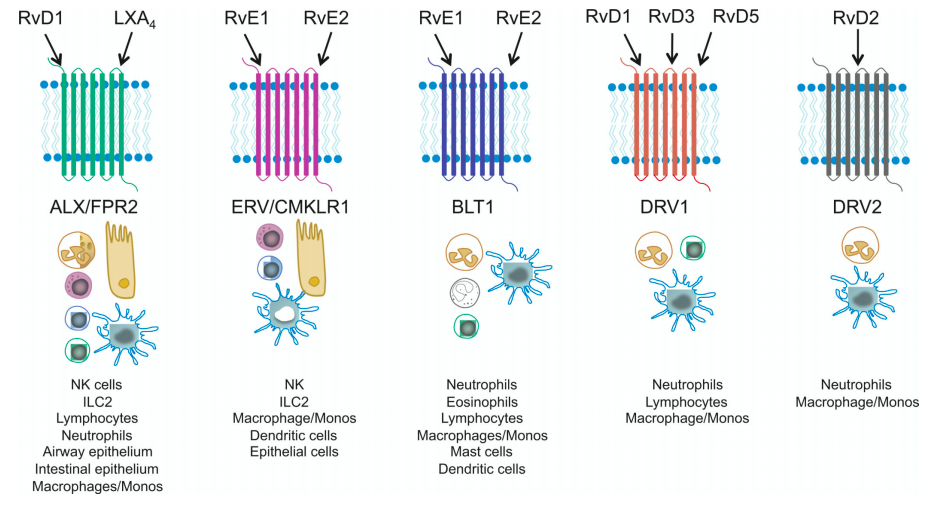
Identification of SPM receptors and their signaling mechanisms
In addition to identification of SPMs, we have also established novel molecular signaling pathways for these mediators by defining their receptors and intracellular signaling mechanisms. In particular, we have focused on polyisoprenyl phosphate remodeling as a key regulator of cell activation. In this work, we have identified a pivotal intracellular signal (i.e., presqualene diphosphate), its regulation of phospholipase D and PI 3-kinase, rapid and transient deactivation by polyisoprenyl diphosphate phosphatase 1, and intersection with receptor-initiated, pro-resolving mediator signaling.
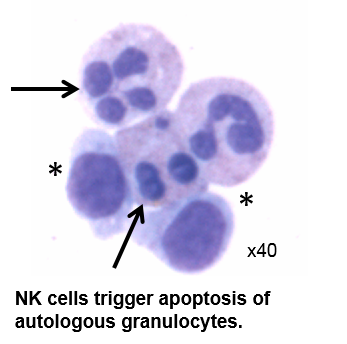
Natural Killer cells, innate lymphoid cells, regulatory T cells and macrophages are cellular effectors of resolution
Using the newly identified pro-resolving mediators as natural resolution “pharmacophores”, we have uncovered pivotal cellular mechanisms that are activated to restrain innate and adaptive immunity in the lung. Current projects in the laboratory are evaluating NK cells, ILCs, Tregs and other cellular mediators in human and murine models of lung inflammation.
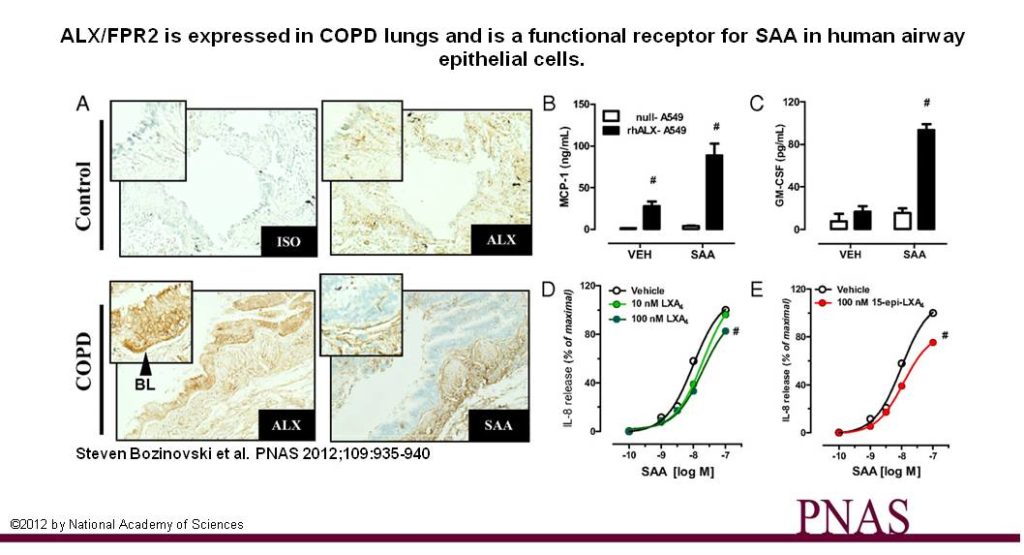
Translation to human pathogenesis
In addition, we are linking basic observations on resolution biology to the genesis of human illness in translational investigations to gain a molecular understanding of pathobiology and to use these insights towards new therapeutic strategies for refractory lung diseases, such as ARDS, severe asthma and COPD. For example, we were the first to show that lipoxins are found in diminished quantities in patients with severe asthma. In contrast, in COPD, we have found that the pro-resolving receptor ALX/FPR2 is pirated by serum amyloid A to stimulate chronic inflammation. Current projects in the laboratory focus on human diseases including asthma and viral lung infection in children.
